| Citation: | Please cite this article as: Jafari Fesharaki M, Ahmadi N, Karimi Taheri K. Reversible heart failure in a patient with hypocalcemic cardiomyopathy. J Geriatr Cardiol 2021; 18(12): 1063−1067. DOI: 10.11909/j.issn.1671-5411.2021.12.009. |
The parathyroid gland is essential to maintaining the serum calcium level within the normal range by secreting the parathyroid hormone (PTH). Significant incidences of increased blood pressure, arrhythmia, left ventricular (LV) hypertrophy, and congestive heart failure (CHF) can occur in patients with parathyroid disease. Hypocalcemia can cause dilated cardiomyopathy, whether this is infrequent. Hypoparathyroidism caused by thyroidectomy and primary hypoparathyroidism are the greatest causes of hypocalcemic heart failure (HF).
The findings from our case may help clinicians to quickly diagnose hypocalcemic cardiomyopathy caused by hypoparathyroidism. Hypocalcemia can lead to severe LV contractile dysfunction irrespective of its cause. The direct action of the PTH on the heart and changes in calcium homeostasis, both hypocalcemia and hypercalcemia, comprise two primary mechanisms whereby disorders of the parathyroid gland affect the cardiovascular system.[1,2] The role of calcium in the myocardium is well-known, and hypocalcemic HF owing to severe vitamin D deficiency, albeit a rare presentation, is well-defined, especially in infants.[3–6]
We herein describe a patient who presented to Department of Cardiology, Shahid Beheshti University of Medical Sciences, Tehran, Iran with acute CHF but without a history of underlying cardiac disease. We found that hypocalcemia and hypoparathyroidism were the potential cause of HF in this patient. A diagnosis of hypocalcemic cardiomyopathy was eventually established, and the symptoms were relieved after the treatment of hypocalcemia, thus, recommend that hypocalcemia and hypoparathyroidism be included in the differential diagnosis of all patients with cardiac dysfunction.
The patient was a 62-year-old man who came to Department of Emergency, Shahid Beheshti University of Medical Sciences, Tehran, Iran with a history of progressive pitting edema in the lower limbs, dyspnea on exertion, and paroxysmal nocturnal dyspnea of one week duration. His past medical history was chronic obstructive pulmonary disease and hypothyroidism with no previous history of cardiac problems. This patient had been diagnosed with hypothyroidism one week earlier and was on levothyroxine (100 μg/d) at hospital admission.
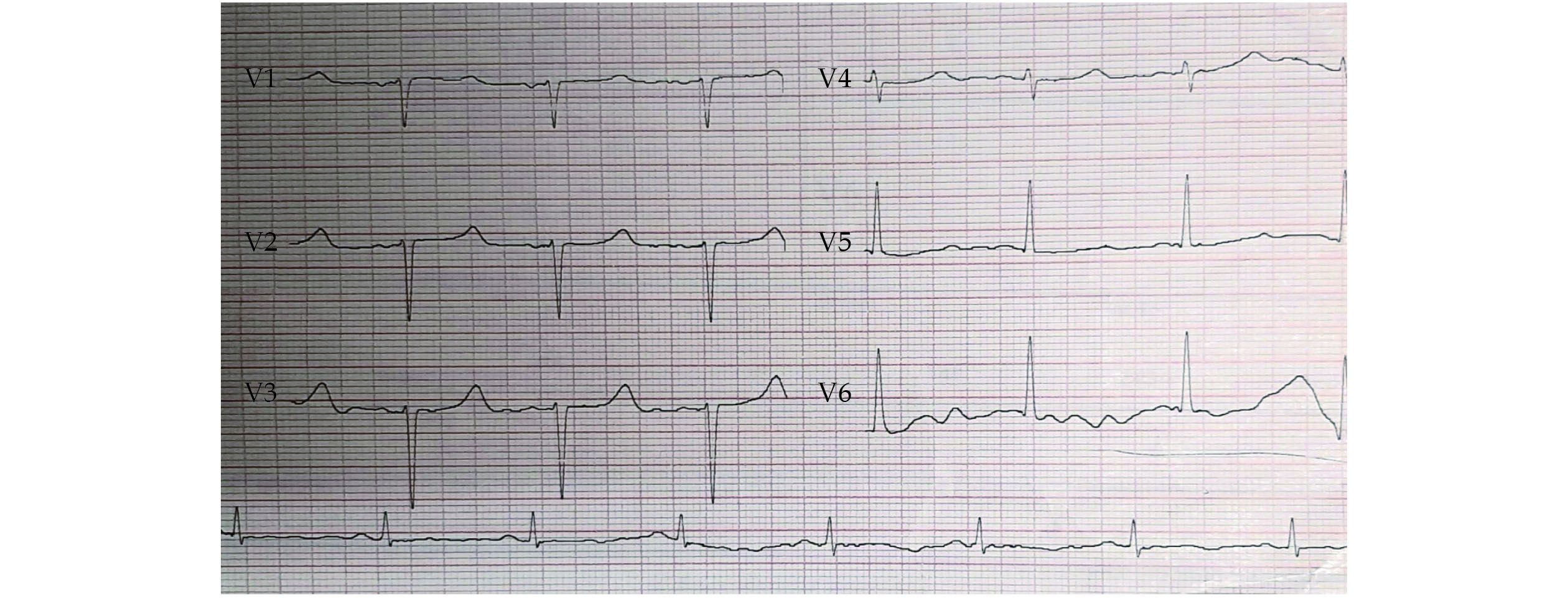
Physical examination revealed a respiratory rate of 18 breaths/min, a heart rate of 60 beats/min, a blood pressure of 120/75 mmHg, an oxygen saturation level of 93% in room air, and a body temperature of 37 ºC. Pulmonary auscultation showed a decreased lung sound at the base of both lungs and diffuse bilateral wheezing. Cardiac auscultation demonstrated normal S1 and S2 without any murmurs. In the examination, the thyroid size was 2 cm × 2 cm without any nodules and tenderness. The Trousseau’s sign was negative, but the Chvostek’s sign was positive. The jugular veins were distended, the peripheral pulses were normal, and the lower limbs were symmetrically edematous. Color Doppler sonography of the lower limbs was normal without any evidence of deep vein thrombosis. The chest X-ray showed signs of pulmonary stasis with hilar enlargement due to the involvement of vascular elements and an increased cardiothoracic index. Computed tomography scanning illustrated nodule-like ground-glass opacity, septal thickening in the basal segments of the right lung, and bilateral pleural effusion. The polymerase chain reaction test for COVID-19 was negative. The 12-lead electrocardiogram (ECG) exhibited a long QT interval. In the first ECG measurement, the corrected QT (QTc) was 0.563 s (normal ≤ 0.44 s) (Figure 1).
Because of the long QT interval in the ECG, heart consultation was requested. Echocardiography revealed an enlarged LV, global hypokinesia, moderate LV systolic dysfunction [ejection fraction (EF) = 40%], moderate LV diastolic dysfunction, mild-to-moderate mitral regurgitation, and mild pulmonary hypertension (systolic pulmonary arterial pressure = 40 mmHg) (Figure 2).
During emergency internment, serum electrolytes and laboratory blood tests disclosed severe hypocalcemia with a total serum calcium level of 5.2 mg/dL (Table 1). Based on the low serum calcium level and echocardiographic findings, a diagnosis of HF due to hypocalcemia was suspected. The administration of two vials of calcium gluconate 10% with an infusion of 500 mL of dextrose water in one hour, followed by eleven vials of calcium gluconate in 1,000 mL of normal saline, was immediately started, which elevated the calcium level to 6.5 mg/dL.
| Laboratory data | First day | Last day before discharge | Normal values |
| Calcium, mg/dL | 5.2 | 8.8 | 8.6−10.3 |
| Phosphorus, mg/dL | 7.2 | 6.3 | 2.6−4.5 |
| Magnesium, mg/dL | 1.8 | 2 | 1.8−2.6 |
| Sodium, mEq/L | 136 | 136 | 134−148 |
| Potassium, mEq/L | 5 | 4.1 | 3.5−5.3 |
| Intact parathyroid hormone, pg/mL | 2.3 | 5 | 15−65 |
| Albumin, g/dL | 3.3 | 4 | 3.5−5.2 |
| Troponin, ng/mL | 0.0023 | 0.002 | < 0.019 |
| Urea, mg/dL | 33 | 37 | 19−44 |
| Creatinine, mg/dL | 1.04 | 0.99 | 0.7−1.4 |
| Bilirubin total, mg/dL | 0.8 | 0.8 | 1.0−1.2 |
| Bilirubin direct, mg/dL | 0.2 | 0.4 | < 0.3 |
| Aspartate aminotransferase, IU/L | 21 | 16 | < 37 |
| Alanine transaminase, IU/L | 31 | 20 | < 41 |
| Alkaline phosphatase, IU/L | 179 | 176 | 80−306 |
| C-reactive protein, mg/dL | 15 | 13 | < 10 |
| Erythrocyte sedimentation rate, mm/h | 43 | 3 | 0−20 |
| Hemoglobin, mg/dL | 7.3 | 10.3 | 13.5−18.0 |
| Red blood cell, 106/uL | 3.5 | 4.2 | 4.5−5.2 |
| Mean corpuscular volume, fl | 67 | 69.7 | 80−96 |
| Mean corpuscular hemoglobin, g/dL | 30.5 | 31.8 | 30−36 |
| White blood cell, 103/uL | 8.2 | 7.9 | 4.0−10.1 |
| Platelet, 103/uL | 239 | 265 | 150−450 |
| Prothrombin time, s | 11.4 | 12 | 8.6−12.6 |
| Partial thromboplastin time, s | 27 | 28 | 20−35 |
| International normalized ratio | 1.11 | 1.21 | 0.9−1.3 |
| Haptoglobin, g/L | 1.82 | − | 0.3−2.0 |
The patient’s hemoglobin level was about 7.3 mg/dL (Table 1), which was less than its normal value (13.5−18.0 mg/dL). A peripheral blood smear showed no basophilic stippling. All rheumatologic markers, including antinuclear antibodies, rheumatoid factor, and anti-double-stranded DNA, were within the normal range. Moreover, the prothrombin time, the partial thromboplastin time, the international normalized ratio, the bilirubin (total and direct) level, the liver function test, and the stool exam were also normal. In abdominal sonography, the splenic span was 112 mm, and the liver had a normal size with normal parenchymal echo. The lead level was requested, and it was within the normal range, too. It was, therefore, concluded that the patient’s anemia was due to HF (anemia of chronic disease).
The patient was admitted to the Cardiac Care Unit, where calcium therapy was continued in tandem with daily checks of serum calcium, magnesium, and phosphorus. Moreover, the patient received one isogroup isoRh pack cell. Initially, the magnesium level was low (1.8 mg/dL). However, magnesium sulfate therapy raised it to the normal range, and it did not have any effect on the serum PTH. After five days, the calcium level rose to 8.8 mg/dL, the hemoglobin level reached 10.3 g/dL (Table 1), and the QTc interval decreased to 0.48 ms (Figure 3).
The patient was discharged after one week with improvements in cardiac symptoms and lower limb edema and with no manifestation of hypocalcemia. Three weeks later, echocardiography was performed, and the results revealed improvements in the LVEF (= 50%) and LV dimensions. For the evaluation of coronary artery disease, a myocardial perfusion scan was done, and it exhibited no significant ischemia.
Primary hypoparathyroidism is described as the abnormally low secretion of PTH. Given the reversibility of some cases of hypocalcemic cardiomyopathy, it is vital to determine the best therapy.[3] In healthy persons with a normal blood calcium level, the reported Chvostek’s sign and Trousseau’s sign are up to 25% and 4%, respectively.[7,8] We routinely use the Chvostek’s sign, which has a sensitivity of 29% and a specificity of 75%,[9] for patients suspected of hypocalcemia.
PTH plays an essential role in cardiac function, not only because it maintains calcium homeostasis but also because it exerts a direct positive effect on cardiac muscles as has been revealed in recent molecular research.[10] Although the accurate mechanisms have yet to be elucidated, hypocalcemia may reduce cardiac contractility, while PTH itself seems to directly influence cardiomyocytes.[11]
Calcium homeostasis is disturbed in hypoparathyroidism. This impairment is associated with poor cardiovascular findings such as cardiomyopathy, CHF, and arrhythmia.[12–14] Also, at an increased risk of cardiovascular disease are patients with nonsurgical hypoparathyroidism and impaired calcium homeostasis.[15,16]
In a previous investigation, patients with chronic hypoparathyroidism had a remarkably higher incidence of cardiovascular disease than subjects without chronic hypoparathyroidism (19.4% vs. 9.5%). Additionally, compared with the non-chronic hypoparathyroidism group, the chronic hypoparathyroidism group had higher incidence rates of atrial fibrillation (6.0% vs. 2.7%), tachyarrhythmia (0.7% vs. 0.4%), myocardial infarction (1.9% vs. 1.3%), coronary artery disease (9.6% vs. 5.3%), HF (5.9% vs. 2.4%), stroke (4.6% vs. 2.4%), and cerebrovascular disease (6.0% vs. 3.0%) (all P < 0.001).[17]
In this case, we reported that three weeks of treatment with calcium supplements augmented the LVEF to almost a near-normal level. Nevertheless, previous studies have reported that the recovery of LV function can take up to six months in patients with HF due to hypoparathyroidism. For instance, in a prior investigation, a 40-year-old woman with a history of thyroidectomy for Graves’ disease was hospitalized for syncope and symptoms of HF. The patient had a long QT interval on ECG and a calcium level of 3.6 mg/dL. It took one year of treatment to improve her symptoms.[3] In another study, following one year of treatment, the symptoms of the patient improved, and LV dilation, which had been depicted by the initial echocardiogram, disappeared.[1]
Hypocalcemia is a rare but treatable cause of cardiomyopathy and should, therefore, be considered in all unexplained cases of LV dysfunction.[17–19] This etiology must be suspected in a patient with signs and symptoms of CHF, as was the case in our patient, who presented with lower limb edema, dyspnea, and LV dysfunction. Early calcium supplementation, usually along with vitamin D, can reduce and even reverse systolic and diastolic dysfunction and conduction abnormalities.[2]
| [1] |
de Oliveira Martins Duarte J, Pestana Pereira PML, Sobral ASG, et al. A rare and reversible case of heart failure: hypocalcemia due to hypoparathyroidism. Clin Case Rep 2019; 7: 1932−1934. doi: 10.1002/ccr3.2397
|
| [2] |
Brown SJ, Ruppe MD, Tabatabai LS. The parathyroid gland and heart disease. Methodist Debakey Cardiovasc J 2017; 13: 49−54. doi: 10.14797/mdcj-13-2-49
|
| [3] |
Parepa I, Mazilu L, Suceveanu A, et al. Hypocalcemic cardiomyopathy: a rare heart failure etiology in adult. Acta Endocrinol (Buchar) 2019; 5: 107−112. doi: 10.4183/aeb.2019.107
|
| [4] |
Moges T, Shiferaw Y, Heye T. Maternal vitamin D deficiency: a Culprit for hypocalcaemia induced myocardial failure in a four-month old infant: a case report from Tikur Anbessa Specialized Hospital, Ethiopia. Ethiop J Health Sci 2017; 27: 299−304. doi: 10.4314/ejhs.v27i3.12
|
| [5] |
Kumar M, Saikia D, Kumar V, et al. Vitamin D deficiency presenting with cardiogenic shock in an infant. Ann Pediatr Cardiol 2011; 4: 207−209. doi: 10.4103/0974-2069.84668
|
| [6] |
Yilmaz O, Olgun H, Ciftel M, et al. Dilated cardiomyopathy secondary to rickets-related hypocalcaemia: eight case reports and a review of the literature. Cardiol Young 2015; 25: 261−266. doi: 10.1017/S1047951113002023
|
| [7] |
Page C, Strunski V. Parathyroid risk in total thyroidectomy for bilateral, benign, multinodular goitre: report of 351 surgical cases. J Laryngol Otol 2007; 121: 237−241. doi: 10.1017/S0022215106003501
|
| [8] |
Sturniolo G, Lo Schiavo MG, Tonante A, et al. Hypocalcemia and hypoparathyroidism after total thyroidectomy: a clinical biological study and surgical considerations. Int J Surg Investig 2000; 2: 99−105.
|
| [9] |
Ahmed S, Saeed F, Rahman F. Severe isolated primary hypoparathyroidism in an adult. J Coll Physicians Surg Pak 2011; 21: 111−112.
|
| [10] |
Kalinowski L, Dobrucki LW, Malinski T. Nitric oxide as a second messenger in parathyroid hormone-related protein signaling. J Endocrinol 2001; 170: 433−440. doi: 10.1677/joe.0.1700433
|
| [11] |
Bansal B, Bansal M, Bajpai P, et al. Hypocalcemic cardiomyopathy: different mechanisms in adult and pediatric cases. J Clin Endocrinol Metab 2014; 99: 2627−2632. doi: 10.1210/jc.2013-3352
|
| [12] |
Shoback DM, Bilezikian JP, Costa AG, et al. Presentation of hypoparathyroidism: etiologies and clinical features. J Clin Endocrinol Metab 2016; 101: 2300−2312. doi: 10.1210/jc.2015-3909
|
| [13] |
Kudoh C, Tanaka S, Marusaki S, et al. Hypocalcemic cardiomyopathy in a patient with idiopathic hypoparathyroidism. Intern Med 1992; 31: 561−568. doi: 10.2169/internalmedicine.31.561
|
| [14] |
Shoback D. Clinical practice. Hypoparathyroidism. N Engl J Med 2008; 359: 391−403. doi: 10.1056/NEJMcp0803050
|
| [15] |
Kim SH, Rhee Y, Kim YM, et al. Prevalence and complications of nonsurgical hypoparathyroidism in Korea: a nationwide cohort study. PLoS One 2020; 15: e0232842. doi: 10.1371/journal.pone.0232842
|
| [16] |
Underbjerg L, Sikjaer T, Rejnmark L. Long-term complications in patients with hypoparathyroidism evaluated by biochemical findings: a case-control study. J Bone Miner Res 2018; 33: 822−831. doi: 10.1002/jbmr.3368
|
| [17] |
Gosmanova EO, Chen K, Ketteler M, et al. Risk of cardiovascular conditions in patients with chronic hypoparathyroidism: a retrospective cohort study. Adv Ther 2021; 38: 4246−4257. doi: 10.1007/s12325-021-01787-7
|
| [18] |
Hurley K, Baggs D. Hypocalcemic cardiac failure in the emergency department. J Emerg Med 2005; 28: 155−159. doi: 10.1016/j.jemermed.2004.06.014
|
| [19] |
Solzbach U, Kitterer HR, Haas H. Reversible congestive heart failure in severe hypocalcemia. Herz 2010; 35: 507−510. doi: 10.1007/s00059-010-3374-7
|
| [1] | Marcus VH Carvalho, Priscila C Kroll, Vinicius N Carvalho. Delayed spontaneous reversibility of left bundle branch block in non-ischemic cardiomyopathy: a case report[J]. Journal of Geriatric Cardiology, 2020, 17(3): 164-168. DOI: 10.11909/j.issn.1671-5411.2020.03.006 |
| [2] | Hee-Eun Choi, Chul Kim, Se-Heum Park, Doo-Il Kim, Ki-Hun Kim, Dong-Kie Kim, Seunghwan Kim, Jino Park. Polymorphic ventricular tachycardia during phase II cardiac rehabilitation in a patient with heart failure: a case report[J]. Journal of Geriatric Cardiology, 2020, 17(1): 64-66. DOI: 10.11909/j.issn.1671-5411.2020.01.003 |
| [3] | Bi HUANG, Yuan-Jing LI, Jian SHEN, Yuan YANG, Gang LIU, Su-Xin LUO. D-dimer level and long-term outcome in patients with end-stage heart failure secondary to idiopathic dilated cardiomyopathy[J]. Journal of Geriatric Cardiology, 2019, 16(8): 621-629. DOI: 10.11909/j.issn.1671-5411.2019.08.005 |
| [4] | Signe Hoi Rasmussen, Karen Andersen-Ranberg, Jordi Sanchez Dahl, Mads Nybo, Bernard Jeune, Kaare Christensen, Sabine Gill. Diagnosing heart failure in centenarians[J]. Journal of Geriatric Cardiology, 2019, 16(1): 1-11. DOI: 10.11909/j.issn.1671-5411.2019.01.008 |
| [5] | Sarah Birkhoelzer, Daniel Stevens, Donah Zachariah, Jackie Taylor, Nigel Rowell, Paul R Kalra. Ivabradine tolerability in heart failure[J]. Journal of Geriatric Cardiology, 2018, 15(11): 708-709. DOI: 10.11909/j.issn.1671-5411.2018.11.007 |
| [6] | Ana Ayesta, Helena Martinez-Selles, Antonio Bayes de Luna, Manuel Martinez-Selles. Prediction of sudden death in elderly patients with heart failure[J]. Journal of Geriatric Cardiology, 2018, 15(2): 185-192. DOI: 10.11909/j.issn.1671-5411.2018.02.008 |
| [7] | Xin GAO, Xue–Dong GAN. Tako-tsubo cardiomyopathy in a radical operation for carcinoma of colon patient with PCI history[J]. Journal of Geriatric Cardiology, 2017, 14(8): 537-539. DOI: 10.11909/j.issn.1671-5411.2017.08.010 |
| [8] | Monika Budnik, Radoslaw Piatkowski, Janusz Kochanowski, Renata Glowczynskai, Dariusz Gorko, Robert Kowalik, Arkadiusz Pietrasik, Grzegorz Opolsk. The oldest patient with takotsubo cardiomyopathy[J]. Journal of Geriatric Cardiology, 2015, 12(5): 588-589. DOI: 10.11909/j.issn.1671-5411.2015.05.014 |
| [9] | Bharathi Upadhya, Mark J Haykowsky, Joel Eggebeen, Dalane W Kitzman. Exercise intolerance in heart failure with preserved ejection fraction: more than a heart problem[J]. Journal of Geriatric Cardiology, 2015, 12(3): 294-304. DOI: 10.11909/j.issn.1671-5411.2015.03.013 |
| [10] | Laura Leto, Mauro Feola. Cognitive impairment in heart failure patients[J]. Journal of Geriatric Cardiology, 2014, 11(4): 316-328. DOI: 10.11909/j.issn.1671-5411.2014.04.007 |
| Laboratory data | First day | Last day before discharge | Normal values |
| Calcium, mg/dL | 5.2 | 8.8 | 8.6−10.3 |
| Phosphorus, mg/dL | 7.2 | 6.3 | 2.6−4.5 |
| Magnesium, mg/dL | 1.8 | 2 | 1.8−2.6 |
| Sodium, mEq/L | 136 | 136 | 134−148 |
| Potassium, mEq/L | 5 | 4.1 | 3.5−5.3 |
| Intact parathyroid hormone, pg/mL | 2.3 | 5 | 15−65 |
| Albumin, g/dL | 3.3 | 4 | 3.5−5.2 |
| Troponin, ng/mL | 0.0023 | 0.002 | < 0.019 |
| Urea, mg/dL | 33 | 37 | 19−44 |
| Creatinine, mg/dL | 1.04 | 0.99 | 0.7−1.4 |
| Bilirubin total, mg/dL | 0.8 | 0.8 | 1.0−1.2 |
| Bilirubin direct, mg/dL | 0.2 | 0.4 | < 0.3 |
| Aspartate aminotransferase, IU/L | 21 | 16 | < 37 |
| Alanine transaminase, IU/L | 31 | 20 | < 41 |
| Alkaline phosphatase, IU/L | 179 | 176 | 80−306 |
| C-reactive protein, mg/dL | 15 | 13 | < 10 |
| Erythrocyte sedimentation rate, mm/h | 43 | 3 | 0−20 |
| Hemoglobin, mg/dL | 7.3 | 10.3 | 13.5−18.0 |
| Red blood cell, 106/uL | 3.5 | 4.2 | 4.5−5.2 |
| Mean corpuscular volume, fl | 67 | 69.7 | 80−96 |
| Mean corpuscular hemoglobin, g/dL | 30.5 | 31.8 | 30−36 |
| White blood cell, 103/uL | 8.2 | 7.9 | 4.0−10.1 |
| Platelet, 103/uL | 239 | 265 | 150−450 |
| Prothrombin time, s | 11.4 | 12 | 8.6−12.6 |
| Partial thromboplastin time, s | 27 | 28 | 20−35 |
| International normalized ratio | 1.11 | 1.21 | 0.9−1.3 |
| Haptoglobin, g/L | 1.82 | − | 0.3−2.0 |